Biofilms and Autoimmunity
Autoimmunity is a major interest of mine. I wrote my first published article on the topic nearly 10 years ago (https://ndnr.com/e-version/apr07/apr07.pdf.html), generally outlining what I still believe today is the major causes of autoimmunity.
Now with more than 10 year of experience in the management, treatment and complete remission of many autoimmune diseases, including Ulcerative Colitis, Crohn’s, Multiple Sclerosis, ALS, Rheumatoid Arthritis, Ankylosing Spondylitis, Lupus, eczema, asthma, allergies, Fibromyalgia and CFS, among many others, I feel that I have a fairly good handle on the process of autoimmunity.
The major challenge presents itself when a patient that has not really read about all of the potential “causes” of autoimmune disease, taking “conventional” pharmaceutical management like methotrexate, wants to reverse their autoimmune disease without doing much work! To that I say …. It’s harder than you think! And in some cases people might just want to stay on their disease modifying anti-rheumatic drugs (DMARDs) for life!
The biggest barrier I see in the proper treatment of autoimmune disease is the cost of testing that I require to assess these conditions properly. The stool testing alone, to quantify facultative anaerobic bacterial imbalances outlined in following article costs roughly $800 CAD. Add a SIBO test and IgG/IgE/IgA food reactivity and the cost increases to over $1000 CAD. These assessments do not even include additional testing for gut barrier dysfunction, advanced autoimmunity screening and cytokine testing to show the degree of reactivity and immune dysfunction occurring in the individual, although, in my opinion these additional tests are certainly not essential in most cases.
None-the-less, the success of treatment, in my opinion, is well worth the costs of testing, as these tests provide something to actually treat the autoimmune condition.
The following article is another excellent example of how conventional medicine may be finally coming around to identifying the causes of autoimmunity and the challenges that biofilms play in the autoimmune process.
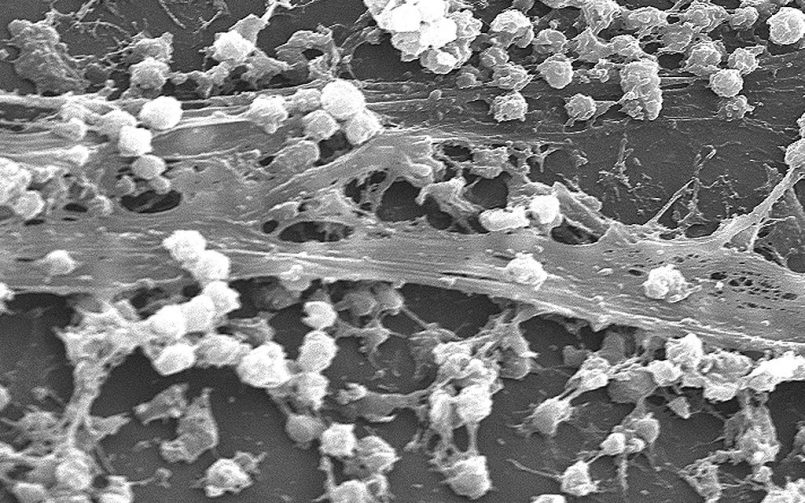
Biofilm is a protective gel composed of proteins, DNA and specific compounds that established bacteria produce to allow them to multiple, while being protected from the harsh ecology of our bodies and the relentless onslaught of our immune systems. As you will read, biofilming is thought to be a very important part of chronic infection and autoimmune disease as these bacteria evade our immune systems, which can trigger an autoimmune disease. Biofilm is also a critical area to focus on in the treatment of any latent bacterial infection associated with autoimmunity, although often treating the infection alone is not always enough to reverse the autoimmune process.
As “normal” bacteria are thought to be the trigger of many peoples autoimmune disease, often the process is not simply to annihilate the bacteria, but requires a combination of bio-terrain balance, immune regulation and removing bacteria from areas where they should not be, such as the small intestine.
Temple-led Research Team Finds Bacterial Biofilms May Play a Role in Lupus
Published: July 6, 2015.
Released by Temple University Health System 
(Philadelphia, PA) – Lupus, multiple sclerosis, and type-1 diabetes are among more than a score of diseases in which the immune system attacks the body it was designed to defend. But just why the immune system begins its misdirected assault has remained a mystery.
Now, researchers at Temple University School of Medicine (TUSM) have shown that bacterial communities known as biofilm play a role in the development of the autoimmune disease systemic lupus erythematosus — a discovery that may provide important clues about several autoimmune ailments.
A team led by TUSM researchers Çagla Tükel, PhD, and Stefania Gallucci, MD, show how bacterial biofilms found in the gut can provoke the onset of lupus in lupus-prone mice. The research is published in the current issue of the journal Immunity. Dr. Tükel is an Assistant Professor of Microbiology and Immunology at TUSM, and Dr. Gallucci is Associate Chair, Microbiology and Immunology, as well as an Associate Professor in Microbiology and Immunology at TUSM. Both are members of the Temple Autoimmunity Center.
“This work stresses the importance of considering infections as a possible trigger for lupus,” Dr. Gallucci said. “Very little was known about how biofilms interact with the immune system because most of the research has been looking at how biofilms protect bacteria, how they make bacteria resistant to antimicrobials such as antibiotics, but almost nothing was known about what biofilms do to the immune response,” she said.
Biofilm is a densely packed bacterial community that excretes proteins and other substances. Those substances form a matrix that protects the bacteria from antimicrobials, the immune system, and other stressors. Biofilms can occur in our guts, among the bacteria that help us digest. They exist as dental plaque, or arise in urinary tract infections. They also can find a home on man-made surfaces such as intravenous catheters. Central to the lupus story is a biofilm protein deposit called an amyloid. In the common gut bacteria E. coli, as well as the bacteria often responsible for severe gastrointestinal distress that accompanies food poisoning, Salmonella Typhimurium, amyloids are called curli because of their curly fiber-like appearance.
Also part of the biofilm is DNA excreted by bacteria. The Temple team discovered that when curli amyloids and DNA meet, they form remarkably durable bonds in the biofilm. When the researchers attempted to separate the DNA from these bonds using a variety of enzymes as well as chemicals, the curli wouldn’t let go. Curli-DNA complexes speed up the creation of the biofilm, the researchers learned. And the Temple researchers found it is also in this composite of curli-plus-DNA that autoimmune trouble appears to arise.
It’s long been known that infection is associated with lupus flares — a flare in lupus is when symptoms worsen. Indeed, infections play a role in between 20 percent and 55 percent of lupus patient mortality. Up to 23 percent of hospitalizations in lupus patients are due to infectious disease complications. Further, the bacteria Salmonella are more aggressive in lupus patients, with the ability to create potentially lethal complications.
The new research shows that the complexes formed from curli amyloid and DNA in the biofilms of both Salmonella and E. coli give rise to not only inflammation, but the self-attacking antibodies of lupus.
To demonstrate the role of biofilms in immune response, the researchers wanted to see how the sentinels of the immune system, called dendritic cells, reacted to a biofilm. The dendritic cells sent “tendrils” into the biofilm and ate up part of it to signal other molecules. Further, they produced large amounts of chemicals called proinflammatory cytokines. These cytokines are important in inciting the immune system to act. Among the cytokines was Type-1 interferon, known to be associated with lupus.
“I was super excited when I saw how activated the dendritic cells were on the biofilm ” Dr. Gallucci said. The levels of cytokines released when dendritic cells were exposed to curli-DNA complexes actually exceeded the most robust response known previously — the response to lipopolysaccharide (LPS).
To test if the immune response seen in the laboratory would be enough to induce autoimmunity and the attack on self that occurs in lupus, the researchers used mice that are prone to develop autoimmune disease. As is the case with many diseases, lupus is the result of a genetic propensity that lies dormant in the absence of an environmental trigger. The researchers wanted to see if the curli-DNA complexes could provide that trigger. They injected susceptible mice with the amyloid-DNA composites or a placebo. Within two weeks, the researchers found the kind of antibodies that attack “self,” known as autoantibodies. The autoantibodies, which target double-stranded DNA, are a diagnostic hallmark of lupus. The response was remarkably fast. It normally takes mice four to five months to develop autoantibodies.
Another strain of mice that do not develop lupus spontaneously but are genetically predisposed to autoimmunity also reacted to the curli-DNA composites with rapid production of autoantibodies. A third strain of mice with no propensity for any autoimmune disease, developed autoantibodies within two weeks of injection, but at lower levels than in the mice with a propensity toward lupus.
All mice developed the autoantibodies whether the curli-DNA composites came from Salmonella or from the kind of E. coli that’s found in a healthy digestive system. In fact, three of the four bacterial families that contain curli genes are found in the gut: Bacteroidetes, Proteobacteria, and Firmicutes, suggesting a possible source of vulnerability in susceptible patients. “How that happens, I think that will be the next level of our project,” Dr. Gallucci said. The research team is already looking at mouse models to see what may lead to the escape of curli-DNA complexes from the gut. Further, the team is collaborating with rheumatologist Dr. Roberto Caricchio, Director of the Temple Lupus Clinic, to see if the patients show signs of exposure to the curli-DNA complexes.
“The next step is to explore the mechanism of how these composites are stimulating autoimmunity,” Dr. Tükel said. “The beneficial bacteria found in our guts can cause problems when they cross the intestinal barrier and reach to places they shouldn’t be. Thus, besides infectious bacteria, a leaky gut could cause many problems. We are now starting to understand how the bacteria in our gut may trigger complex human diseases including lupus. So it’s critical for us to understand the biology of the bacterial communities and their interactions with the immune system.”
The research may offer clues to diseases involving amyloids, Dr. Tükel said. For instance, amyloid plaques in the brain are a signature of Alzheimer’s disease. Type-2 diabetes and Parkinson’s disease also feature amyloids. “Right now, we can only speculate,” she said. “Some reports suggested that antibiotic treatment may be changing the course of Alzheimer’s disease. Antibiotics are thought, in some cases, to slow the mental decline suggesting that bacterial infections, or a bacterial component, may be contributing to the disease. That could be a link, but for the moment, it’s only speculation.”
Understanding how biofilms trigger autoimmunity may ultimately lead to changes in patient treatment, Dr. Gallucci said. “So understanding how the biofilms affect flares could lead to a different treatment approach. Now, they give immune suppressive drugs. Maybe you want to do something else, like treat the underlying infection.”
You must be logged in to post a comment.